Native starches, while versatile, often fall short in meeting the rigorous demands of modern industrial applications. They can be sensitive to heat, acid, and shear, or may lack the necessary stability for freeze-thaw cycles and extended shelf life . Modified starches are engineered to overcome these limitations. By altering the physical and chemical structure of the starch granule, manufacturers can create tailor-made ingredients with specific functional properties. This article provides an overview of the primary processing techniques used to produce modified starch, categorized by the nature of the modification.

1. Physical Modification
Physical modifications are often favored for their simplicity and for producing “clean label” ingredients, as they involve no chemical reagents .
- Pre-Gelatinization: This is one of the most common physical methods. The starch is cooked (gelatinized) in water and then dried, typically on a drum dryer or via extrusion. The resulting product can swell and thicken in cold water without heating, making it ideal for instant foods.
- Heat-Moisture Treatment (HMT) and Annealing: These are hydrothermal treatments that modify the physicochemical properties of starch without destroying the granule structure.
- Heat-Moisture Treatment (HMT): Starch with a low moisture level (typically less than 35%) is treated at high temperatures (e.g., 80-140°C) for a set period . This process alters the crystalline structure, often increasing the gelatinization temperature and improving thermal stability.
- Annealing: This involves treating starch in excess water (at least 40% moisture) at a temperature below its gelatinization point. Annealing perfects the crystalline structure within the granule, leading to a sharper gelatinization temperature range and increased stability.
- Physical Modification with Small Molecules: An emerging technique involves hydrothermally treating starch in the presence of small molecules (like sugars or sugar alcohols). This clean-label method can precisely tune properties such as gelatinization temperature and viscosity, offering a broader range of functional outcomes than traditional hydrothermal treatments alone .
2. Chemical Modification
Chemical modification is a powerful and widely used method that introduces functional groups into the starch molecule via reactions with various chemical reagents. The goal is to alter hydrogen bonding, add steric hindrance, or introduce ionic charges to change how the starch behaves in a system.

- Esterification and Etherification: These are two of the most common forms of chemical modification.
- Esterification: This involves reacting the hydroxyl groups on the starch molecule with an acid or acid derivative. A common example is the production of starch phosphates (using salts like sodium trimetaphosphate or sodium tripolyphosphate), which are cross-linked to provide high viscosity and stability under heat and shear . Another common esterification is with citric acid, which forms ester bonds (citrate starch), increasing rigidity and reducing digestibility .
- Etherification: This reaction introduces ether groups, such as in hydroxypropylated starch. This modification improves clarity, reduces the gelatinization temperature, and enhances freeze-thaw stability by preventing the retrogradation (re-crystallization) of amylose molecules.
- Cross-Linking: This is a specific type of esterification or etherification where a bifunctional reagent (like phosphorus oxychloride or sodium trimetaphosphate) creates chemical bridges between different starch molecules. Cross-linking reinforces the granule, making it highly resistant to high temperatures, acidic conditions, and mechanical shear.
- Acid Hydrolysis: In this classic process, starch is treated with a dilute acid (e.g., hydrochloric acid) at a temperature below its gelatinization point . The acid preferentially attacks the more amorphous regions of the granule, cleaving long polysaccharide chains into shorter ones. This results in “acid-thinned” starches that form strong, rigid gels when cooked, widely used in confectionery like gum candies. The process can be enhanced with a preceding steam explosion to increase solubility and reduce crystallinity .
The diagram below summarizes the relationship between the main types of modification processes and their resulting functionalities:
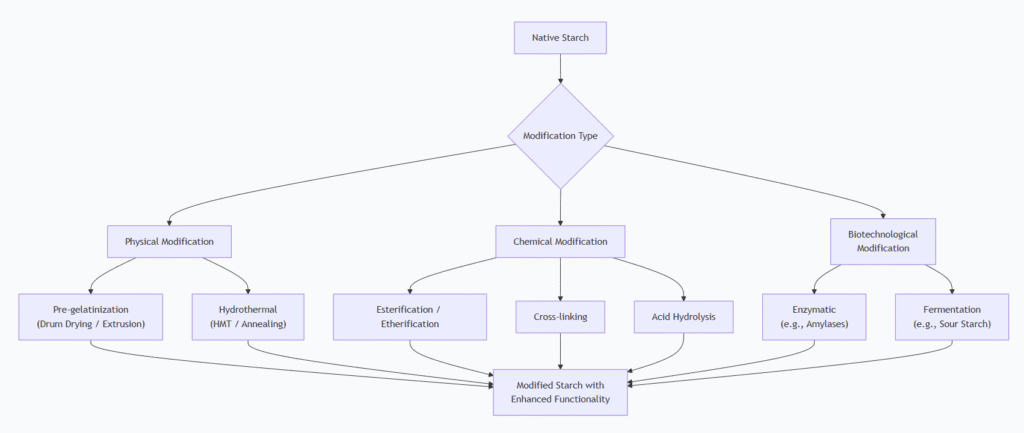
3. Biotechnological and Enzymatic Modification
These methods leverage biological systems to achieve specific modifications, often with high precision and under milder conditions.

- Enzymatic Modification: Enzymes offer a highly specific way to modify starch structure. For example, α-amylase can be used to randomly hydrolyze α-1,4 glycosidic bonds, reducing viscosity and producing maltodextrins . Other enzymes, like pullulanase, specifically cleave the branch points (α-1,6 linkages) in amylopectin to create a linear starch with a high amylose-like character, which is excellent for forming strong films.
- Fermentation: This traditional method, used for centuries to produce products like sour cassava starch, relies on the action of naturally occurring or selected microorganisms . During fermentation, microorganisms such as Bacillus species produce organic acids and enzymes (like amylases) that hydrolyze the starch . This process modifies the starch’s pasting properties and expansion capability, a key trait for specific baking applications . Researchers are now developing standardized, scalable fermentation processes to replace traditional, empirical methods .
4. Other Specialized Techniques
- البثق التفاعلي: This combines the chemical reaction and the extrusion process into a single, continuous step. Starch, reagents (e.g., citric acid for esterification, cross-linking agents), and often a plasticizer are fed into a twin-screw extruder . The high temperature, pressure, and intense mechanical shear within the extruder facilitate both the chemical reaction and the gelatinization of the starch, producing a modified thermoplastic starch in one efficient operation .
- Gamma Irradiation: This physical method uses ionizing radiation to generate free radicals on the starch molecules. These radicals can cause chain scission (degradation), leading to reduced viscosity and increased solubility . While effective, high doses can cause instability, and it is sometimes combined with other agents, such as inorganic peroxides, to achieve both low viscosity and good stability at lower radiation levels .
In conclusion, the production of modified starch is a diverse and sophisticated field. The choice of process—whether physical, chemical, or biotechnological—is dictated by the desired functional properties and the target application, allowing starch to be transformed from a simple raw material into a high-performance industrial ingredient.



